Indice degli argomenti
Digital Therapeutics is a subset of digital health where the medical treatment is based on the use of software, or software-driven solutions, in conjunction with hardware devices and traditional medical therapies such as drugs. Digital therapeutics are clinically validated treatments for the cure of many diseases showing to be particularly effective in pathological conditions related to both behavioural and psychological factors that can benefit from real-time monitoring of behaviour and state, such as Alzheimer’s disease, dementia, type II diabetes, serious respiratory problems such as chronic obstructive pulmonary disease, asthma, gastrointestinal problems, obesity, substance abuse, ADHD, insomnia, hypertension, anxiety, depression, autism, cardiovascular diseases. Many pathologies addressed by digital therapeutics, such as chronic diseases or neurological disorders, are now poorly handled by national health systems because they are quite expensive. Digital therapies are able to provide cheaper treatment than traditional therapies, reducing the demand for medical care.
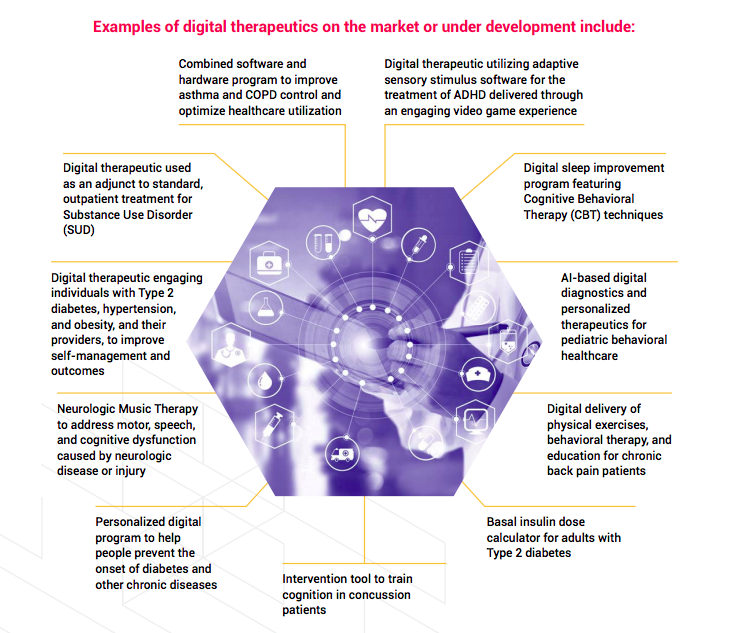
Digital Therapeutics examples
Digital therapeutics are software, we said, for the treatment of specific diseases. Sometimes, they are a substitutive therapy (or the only therapy) acting with sensory stimuli; in other cases, they help or improve traditional medicine combining the use of software with devices or drugs. For example, some devices may complement traditional treatment by helping patients in managing and monitoring their conditions, offering indications, like remembering them when and how much medicine to take.
Digital Therapeutics: a new era of medicine
The global market for digital therapeutics is expected to reach $9 billions by 2025 (source: Crystal Market Research). Factors driving the market growth are, besides the broad benefits, the rising incidence of chronic diseases such as diabetes, the increased focus on health prevention, technological advancements, the need to restrain health costs, the jump in venture capital investments in this field. Digital therapeutics have to follow a path of clinical validation and quality standards, as for real drugs, to establish themselves as a form of proper, effective and viable treatment and thus be recognized by the medical community, regulators, patients to enter the sphere of health treatments covered by public health services or insurance companies. Among the advantages of digital therapeutics are a reduction in health costs and the empowerment of patients towards the daily care of their health. 
Digital Therapeutics, a field of collaboration between startups and large companies
Digital therapeutics embody a new age of care and cure that sees contamination between the software and pharma industries. In this field, there is strong cooperation between startups and large companies, many of which founded in 2017 an international non-profit organisation called Digital Therapeutics Alliance. The Digital Therapeutics Alliance brings together the leading manufacturers of clinically validated digital therapeutic solutions (DTx) in the healthcare industry. Its mission is both to broaden the understanding, adoption and integration of digital therapeutic solutions in traditional healthcare; and also promote principles, standards in products and protocols, high quality, clinical validation, usability, safety (including data), effective adherence to the real needs of the patient (including through their involvement in development processes), respect for privacy, sharing of test results and advances in research. To deepen the principles and the point of view of the association, read the White Paper. In Europe, an organization working to support the digital therapeutics sector is EIT Digital, with its area dedicated to digital wellbeing. EIT Digital is a leading European organization for innovation and education in digital entrepreneurship, realizes acceleration programs and connects a pan-European ecosystem of more than 180 large companies, deep tech scaleups and startups, universities and research institutes in Europe, where students, researchers, engineers, engineers, entrepreneurs and entrepreneurs work together in an open innovation context. Much attention goes to digital solutions for young people to prevent or detect early mental and physical problems (e.g. depression, overweight, obesity, chronic diseases). There are several interesting companies in this area that EIT Digital is supporting in their path (which also includes clinical validation), for example, ACT4Y (Asthma Control For You) which aims to enable asthmatic patients to achieve better results, thanks to intelligent inhalers and the use of data to customize and ‘calibrate’ the use of drugs. Asthma is a disease that affects over 300 million people worldwide, a number that is set to rise to 400 million by 2025.
Digital Therapeutics: a case study
In the last decade, digital therapeutics have made significant progress, using technology to complement or replace traditional clinical therapy. The digital therapeutics sector is currently far ahead in the United States where the regulatory system is helping to grow it through the FDA. FDA has already embraced digital health and digital therapeutics with programs and guidelines, setting standards and offering the possibility for companies in the sector to apply for and obtain (if deemed valid) approval of their solution as a health treatment that can be prescribed and reimbursed by the national health service. The first digital therapeutic that received such approval was reSET – a software-only treatment for opioid addiction – developed by the startup Pear Therapeutics in collaboration with Sandoz, a Novartis’ division. reSET is essentially an app that applies cognitive behavioural therapy (CBT) to help outpatients living with these addictions. The two companies are also collaborating on the development of applications for schizophrenia and multiple sclerosis. Below is the Pear Therapeutics PDT product development pipeline where Novartis/Sandoz is involved. 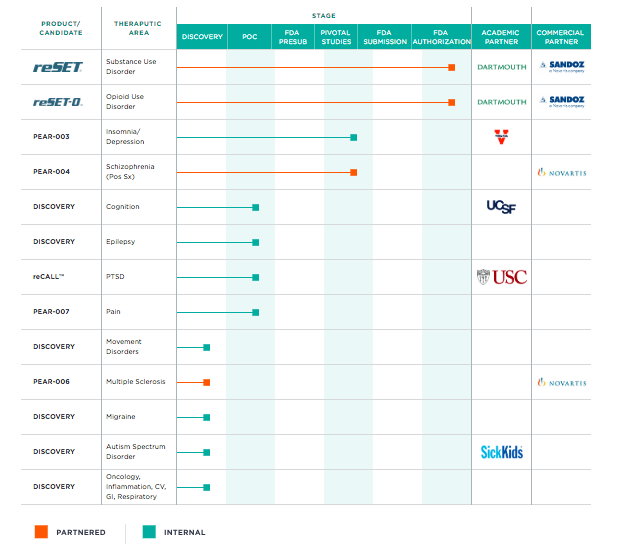
Digital Therapeutics startups worldwide
In addition to the mentioned Pear Therapeutics, several startups are working in digital therapeutics with extreme expertise and intent in satisfying therapeutic needs often faced with superficiality by national health systems, although they can also become disabling for the person. Therapies for depression or addiction, diseases such as anxiety and insomnia, which can have a tremendous impact on the overall well-being of the person, are usually minimised and can result in great bias in the context of social life. These startups typically have rather large and multidisciplinary teams, including software specialists (developers, data scientists, artificial intelligence experts, gaming specialists, etc.), health specialists (doctors, researchers, pathology specialists, psychologists, behavioural therapeutic) and managers who can interact with large companies, pharmacies, administrations. Very often they have academic partners and advisors. All companies start from a product but are structured to become platforms, capable of developing a pipeline of products. They are the new pharma companies.
- Akili Interactive – They’re bringing together world-class neuroscience with exceptional creativity and video game development to challenge the status quo of medicine;
- Propeller Health – Propeller creates products to more effectively treat chronic respiratory disease and improve clinical outcomes for patients through connectivity, analytics, and companion digital experiences;
- WellDoc – Welldoc’s award-winning digital therapeutic, BlueStar, is a digital assistant for people with type 2 diabetes;
- Big Health– Big Health’s purpose is to help millions back to good mental health, which it does by creating fully automated and highly personalized behavioural programs for mental health;
- Sornormed – They developed Tinnitracks, a web application which allows you to filter your music in order to use it for a new tinnitus therapy that is endorsed by the latest neuroscientific findings;
- Kaia Health, they develop AI for Digital Therapeutics, they already sell an App that coaches back-pain sufferers how to correctly perform therapeutic exercises known to provide relief;
- Proteus Digital Health – They developed Proteus Discovery, a new system that measures medication treatment effectiveness, helps physicians improve clinical outcomes and patients reach health goals. It comprised of ingestible sensors, a small wearable sensor patch, an application on a mobile device and a provider portal;
- Tilak Healthcare – Tilak is a unique videogame studio where like-minded & passionate people create fun mobile medical games to monitor patients with chronic diseases;
- Click Therapeutics – Click Therapeutics develops and commercializes software as prescription medical treatments for people with unmet medical needs, leveraging on cognitive and neurobehavioral mechanisms; their main solution so far is Clickotine, a personalized smartphone app for smoking cessation;
- Voluntis – They develops companion software for the healthcare sector, in particular solutions for diabetes, oncology and blood problems such as hemophilia. Voluntis was the first digital therapeutics company to be listed on the stock exchange in May 2018 (press release);
- Amiko – Founded in Italy in 2015, Amiko develops devices for patients with chronic diseases that track the use of drugs and transfer data to a platform that allows you to verify the correct assumption. Amiko’s flagship product is Respiro, a digital solution that allows advanced monitoring of the use of respiratory drugs to improve adherence and effectiveness of treatment.
Donatella Cambosu
© RIPRODUZIONE RISERVATA